

The variations in the structure of Ca-aluminosilicate melts as a function of composition have measurable effects on their macroscopic thermodynamic properties. Recent studies indicate that volumetric properties do not follow a linear dependence on composition in the system CaO-Al2O3-SiO2, especially along the Ca3Al2O6-SiO2 join. In an attempt to derive possible correlations between the geometrical configuration of the silicate framework and the observed variations in the physical and chemical properties, we have engaged in a study of the determination of the structure of glasses in the system CaO-Al2O3-SiO2 by X-ray absorption spectroscopy. In particular, we have addressed the problem of the effect of variable Al/Si ratio on the Al site geometry. Changes in the geometry of the Al polyhedra are believed to significantly affect the volumetric properties of the investigated glasses. The K-edge spectra are characterized by a main absorption band composed of two peaks of variable intensity at about 1567 eV and 1570 eV. The energy positions of these two peaks do not shift significantly as the composition changes whereas their relative intensities exhibit a distinct variation, with the 1567 eV peak decreasing in intensity and the 1579 eV peak increasing as the silica content decreases (SiO2 varies from 65 mol% in the silica-rich member to 9 mol%). Empirical correlations between the glass spectra and those of crystalline compounds indicate that the peak near 1567 eV is characteristic of Al in tetrahedral coordination whereas VI-fold coordinated Al is revealed by the shift of the main edge up to 1570 eV. The appearance of two peaks (in unequal and variable proportions) is therefore qualitatively consistent with the presence of Al residing in both tetrahedral and octahedral sites in the Ca-aluminosilicate melts.
In order to obtain more quantitative information on the effect of coordination
and distortion of the Al site from the XANES features, we performed XANES
simulations according to one-electron multiple scattering (MS) theory.
Muffin-Tin potentials with X-alpha exchange terms were constructed for
the various clusters. Tetrahedral and octahedral Al theoretical clusters
(5 and 7 atoms, respectively) have been combined in different proportions
(25 % O + 75 % T; 20 % O + 80 % T; 13 % O + 87 % T; 5 % O + 95 % T). The
effect produced as a function of the increase of the tetrahedral component
is that the resonance at lower energy increases in intensity, whereas the
one at higher energy decreases. The trend in the calculated spectra reproduce
very well that found in the experiments (Fig. 3.8-6), indicating the presence
of both four-fold and six-fold coordinated Al in all the samples investigated.
The decrease in the SiO2/Al2O3 ratio is
accompanied by a relative stabilization of six-fold Al in the glass. Preliminary
calculations on octahedral and tetrahedral clusters show also that both
symmetrical and asymmetrical distortion as well as variation in Al-O bond
distance produce only small differences in the chemical shift and in the
intensities of the two absorption peaks. Structural models involving local
change in Al environment can shed some light on the interpretation of the
non-linear composition dependence of macroscopic properties such as molar
volume and compressibility in the ternary system CaO-Al2O3-SiO2.
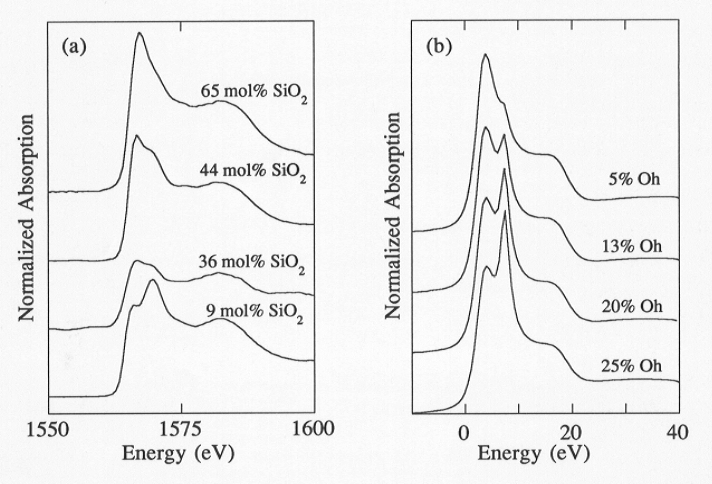 |
Fig. 3.8-6: (a) Experimental XANES spectrum of Al K-edge along the join SiO2-Ca3Al2O6. The relative intensity of the two peaks at about 1567 and 1570 eV changes as a function of the composition of the system. (b) Linear combination of tetrahedral and octahedral Al theoretical spectra. The weighted sum of AlIV and AlVI spectra (% OH refers to % Al in octahedral coordination) in different proportions reproduce very well the relative intensity change observed in the experimental spectra. |

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page