

Pigeonites are calcium-poor monoclinic pyroxenes that are important components of volcanic and plutonic rocks. The chemistry, state of order and especially microstructure of exsolved phases of pigeonites can often be used as an indication of the cooling rate of the rock. Such estimates would be greatly improved by more complete thermodynamic data on the Ca-poor clinopyroxene phase. Naturally-occurring pigeonites contain up to 10 mol% Ca substitution for Mg and Fe in the formula (Ca,Mg,Fe)SiO3, along with other relatively minor chemical substitutions. However, these additional chemical substitutions, together with the possible presence of oxidised Fe3+ and exsolved Ca-rich clinopyroxenes and/or orthopyroxenes argues against the use of such natural material for the calibration and measurement of solid solution properties of the Ca-poor clinopyroxenes. As an initial step towards providing complete thermodynamic data for the (Ca,Mg,Fe)SiO3 system, we have therefore undertaken a study of the volume variation with composition of synthetic Ca-free clinopyroxenes.
At pressures below about 7 GPa Ca-poor clinopyroxenes are only stable
at relatively low temperatures, below ~ 700
- 800 oC. Such temperatures are too low to allow reasonable
crystal growth on a laboratory time-scale, so previous syntheses of (Mg,Fe)SiO3
clinopyroxenes have therefore been carried out metastably at higher temperatures
within the stability field of orthopyroxene. This method runs the risk
of synthesising clinopyroxenes with significant densities of stacking faults
whose local structure resembles that of orthopyroxene and which could consequently
influence the measured thermodynamic properties. We have therefore taken
advantage of the fact that orthopyroxenes transform to high-clinopyroxenes
at temperatures and pressures in excess of ~
1000 oC and 7 GPa, as reported for MgSiO3 in last
year's Annual Report. Syntheses were performed with the multi-anvil presses
at a range of temperatures and pressures above this phase boundary, and
therefore in the stability field of high-clinopyroxene. Upon pressure release
in the multianvil press these high-clinopyroxenes invert to the low-clinopyroxenes
of interest. Low clinopyroxenes of fourteen different compositions on the
join between MgSiO3 and FeSiO3 were synthesised in
this way, and the cell parameters determined by X-ray powder diffraction.
The most important thermodynamic result from this study is that there is
no detectable excess volume of mixing between MgSiO3 and FeSiO3
(Fig. 3.3-1), while the ß angle of the monoclinic
unit cell shows a significant maximum at around Mg0.43Fe0.57SiO3
(Fig. 3.3-1). The crystal-chemical reason for this behaviour remains to
be determined.
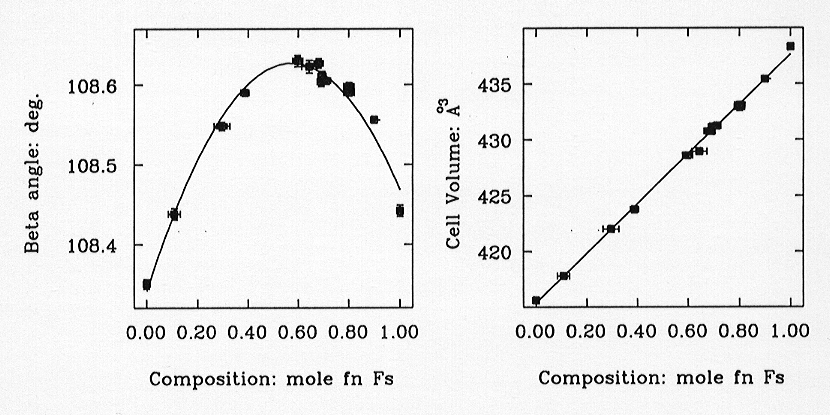 |
Fig. 3.3-1: The variation of the unit-cell ß-angle (left) and volume (right) of low-clinopyroxenes with space group P21/c with composition across the join MgSiO3 (=Fs0.0) to FeSiO3 (=Fs1.0). |

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page