

Alkali feldspars crystallized from felsic magmas with high iron content commonly contain more than 1 wt% of Fe2O3. Preliminary hydrothermal experiments on pantelleritic melts at high oxygen fugacity have shown that more than 5 wt% of Fe2O3 can be accommodated in alkali feldspar under magmatic conditions as a result of increases in oxygen fugacity for these normally very reduced magmas. It is therefore apparent that the substitution of ferric iron into alkali feldspar is potentially of great importance in controlling the abundance and oxidation state of iron in iron-rich felsic liquids. This important substitution has received little attention in the past, and we have consequently undertaken to characterize the substitution mechanism and map out a solvus in the system KAlSi3O8-KFeSi3O8.
We have successfully synthesized the endmember KFeSi3O8 in two ways. In one set of experiments the components of the iron feldspar were added as oxides and carbonates to a K2O-V2O5 flux at one atmosphere and 700 °C, and allowed to stand for one month in a Pt crucible. The crucible was quenched in air, yielding a vanadate glass containing silicate crystals. The vanadate glass was dissolved in water in an ultrasonic bath, leaving a residue of silicate crystals 100 mm to 1 mm in size. Electron microprobe and single crystal X-ray diffraction analysis showed predominantly KFeSi2O6 with the leucite structure. Very small quantities of KFeSiO4, apparently with kalsilite structure, and KFeSi3O8 were also found as fine-grained encrustations around unreacted Fe2O3 powder.
In the second series of experiments, powdered glass with a nominal composition
of KFeSi3O8 was loaded with a drop of distilled H2O
into 3mm Pt capsules and welded shut. The capsules were then placed in
5mm Au capsules with sintered Fe2O3 and a drop of
water. These double capsules were run in a 3/4" piston-cylinder apparatus
with a talc-pyrex cell at a nominal pressure of 7.5 kbar (using 22 % friction
correction). A run at 650 °C yielded glass; two runs at 550 °C
yielded mixtures of a reddish opaque powder and clear, pale greenish to
colourless crystals. A single-crystal X-ray diffraction study on one of
these crystals indicated that it has unit-cell parameters a = 8.664(3)Å
, b = 13.110(4) Å , c
= 7.331(2) Å , α
= 90.75(2)°, ß = 116.08(2)°, γ
= 86.57(3)°, and space group C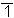 bar. A
structure refinement based on 2164 measured reflection intensities confirmed
the structure type of the crystal as being that of microcline. Average
tetrahedral bond lengths of T1o = 1.85Å
, T1m = 1.61Å , T2o = 1.61Å
, T2m = 1.62Å indicate that Fe3+
is essentially completely ordered onto the T1o site which is the Al site
in KAlSi3O8 microcline.
bar. A
structure refinement based on 2164 measured reflection intensities confirmed
the structure type of the crystal as being that of microcline. Average
tetrahedral bond lengths of T1o = 1.85Å
, T1m = 1.61Å , T2o = 1.61Å
, T2m = 1.62Å indicate that Fe3+
is essentially completely ordered onto the T1o site which is the Al site
in KAlSi3O8 microcline.
If these results are applicable to low levels of Fe3+ substitution
in microcline, then the associated configurational entropy (of the direct
substitution of Fe3+  Al3+
on the T1o site) will be significantly less than that which would be calculated
for a full random-site substitution. Future experiments will explore this
tentative conclusion by attempting to synthesize further feldspars of intermediate
compositions along the join KFeSi3O8 - KAlSi3O8.
Al3+
on the T1o site) will be significantly less than that which would be calculated
for a full random-site substitution. Future experiments will explore this
tentative conclusion by attempting to synthesize further feldspars of intermediate
compositions along the join KFeSi3O8 - KAlSi3O8.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page