

Previous investigations of the dependence of the solubility of Ni in silicate melts on temperature- and oxygen fugacity, together with spectroscopic investigations of Ni-bearing silicate melts, have raised the question of a potential compositional dependence of Ni solubility.
Three separate experiments have been performed using the mechanically assisted equilibration method. Each experiment involved the stepwise mechanical addition of a chemical component to the melt, followed by equilibration, at constant temperature and fO2. SiO2, Mg2SiO4 and Na2SiO3 were added in separate experiments. In all experiments performed, oxygen fugacity was controlled by 1:1 CO/CO2 gas mixtures.
The addition of SiO2 at 1350 °C and log fO2 = - 9.14 had no influence on the solubility of Ni up to the maximum amounts investigated corresponding to 30 wt% SiO2 addition. The addition of forsterite (Mg2SiO4), at 1400 °C and log fO2 = - 8.6, also revealed no effect on Ni solubility.
Up to 45 wt% Na-metasilicate (Na2SiO3) was added at 1300 °C and log fO2 = -9.7. This temperature was chosen to keep the loss of Na2O by volatilisation as small as possible during the long experimental run duration. The initial Ni solubility decreased strongly with increasing amounts of Na-metasilicate up to about 22 wt% from 2450 to 1800 ppm. Further addition of Na-metasilicate then led resulted again in an increase of Ni solubility to about 2500 ppm at 45 wt% Na-metasilicate content in the melt.
This unusual solubility behaviour of Ni can be explained by a dual role
of Ni in the structure of the melt. In the initial addition of Na2SiO3
component to the melt the added alkali exchanges with Ca and Mg to charge
balance the network aluminum. That is, the Na replaces the Ca and Mg as
a network stabilizing cation. The excess Ca and Mg decrease the Ni solubility
as has been demonstrated in previous work. When all the aluminum has been
charge balanced by Na (at a mole ratio of Na/Al = 1:1) the solubility increases
again because further addition of Na creates a networkmodifying alkali
unit within the melt structure that has a high affinity for Ni. The potential
for metal solubilities to reveal such structural subtleties in silicate
melts holds significant promise for the future of such investigations.
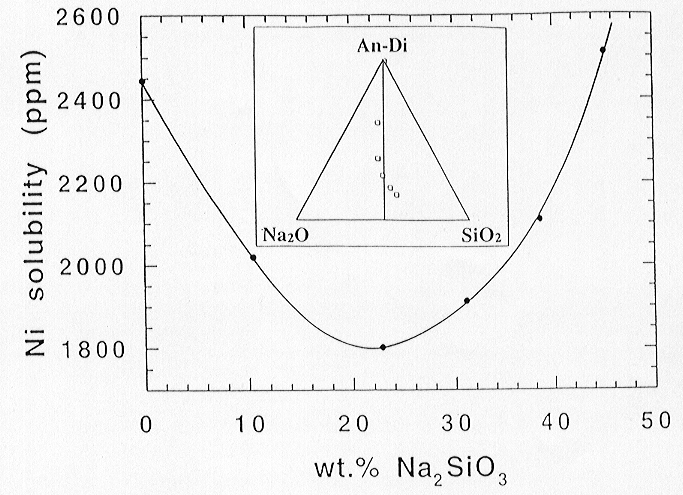 |
Fig. 3.2-4: Solubility dependence of Ni on the amount of added Na2SiO3 to the melt (in wt%). The corresponding melt composition in the system Na2O-SiO2-An-Di is indicated separately in the inserted diagram. |

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page